
Federal Register notice: FDA announces a 6/16 Oncologic Drugs Advisory Committee meeting of its Pediatric Oncology Subcommittee that will discuss dosa...

Federal Register notice: FDA releases a final guidance on drug testing for high-risk drug components.

Federal Register notice: FDA announces the revocation of three emergency use authorizations (EUAs) for Covid-19 tests because they are no longer being...

CDER director Patrizia Cavazzoni says the Center is working with CBER and CDRH on discussion papers involving artificial intelligence/machine learning...

FDA medical reviewers question the effectiveness and safety of Sareptas SRP-9001 as a treatment for some cases of Duchenne muscular dystrophy and ask ...

Three congressional Democrats introduce legislation to require drug companies to accurately account for how much they spend on drug research and devel...
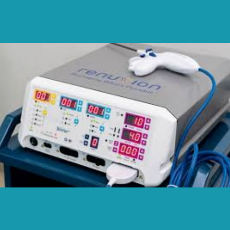
FDA clears the Apyx Renuvion/J-Plasma handpiece for coagulation of subcutaneous soft tissues following liposuction for aesthetic body contouring.
FDA approves a Chiesi Global Rare Diseases and Protalix BioTherapeutics BLA for Elfabrio (pegunigalsidase alfa-iwxj) for treating adult patients with ...