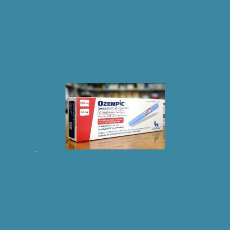
FDA says it has seized thousands of units of counterfeit Ozempic that also contain counterfeit needles.

FDA commissioner Robert Califf says a new final rule allowing some waivers of informed consent will enable minimal-risk research that would not otherw...

FDA publishes a draft guidance revising a recent draft on quality considerations for topical ophthalmic drugs.

FDA warns Sarasota, FL-based Omeza about CGMP violations in its production of unapproved and misbranded over-the-counter drugs.

FDA issues a complete response letter for the Merck resubmitted NDA for gefapixant, saying the drug did not establish substantial evidence of efficacy...

Medline Industries recalls some lots of tracheostomy care and cleaning trays following complaints.

FDA says Insight Pharmaceuticals is recalling one lot of its Americaine benzocaine topical anesthetic spray due to the presence of benzene from the ca...

FDA warns Brooklyn, NY-based Hua Da Trading that it is illegally marketing a misbranded unapproved drug containing the active ingredient in Viagra as ...