
Federal Register notice: FDA makes available a draft guidance entitled Good Manufacturing Practice for Active Pharmaceutical Ingredients (APIs) Used i...

CBER says Lewisburg, TN-based Vitacell Biologics is illegally marketing cellular and exosome products.
FDA releases the form FDA-483 with two inspection observations issued to Columbus, OH-based STAQ Pharma of Ohio.
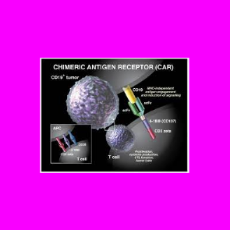
CBER executives say FDA is aware of a relatively small number of cases in which people treated with CAR-T products developed secondary cancers that ma...

FDA issues a finalized guidance on updating ANDA labels following revisions to the reference-listed drug label.

CDER and CBER say they have accepted the first artificial intelligence, digital health technology, and neuroscience project in the ISTAND pilot for de...

FDA publishes a comprehensive Veterinary International Conference on Harmonization guidance on good manufacturing practice for animal drug APIs.

FDA purchases a subscription to digiM Solutions I2S software for the CDER Division of Product Quality and Research.