
FDA sends PharmaTher a complete response letter on its ANDA for Ketarx (ketamine) and its use in anesthesia, sedation, and pain, among other uses.
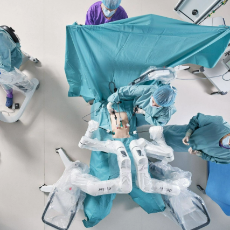
FDA grants Distalmotion a de novo marketing authorization for the Dexter Surgical Robot for adult inguinal hernia repair.

Kinova recalls (Class 1) its Jaco assistive robotic arm to correct the device due to an increased fire hazard.

FDA issues a safety alert recommending that healthcare providers not use BioZorb Markers and BioZorb LP Markers manufactured by Hologic.

FDA approves an Iterum Therapeutics NDA for Orlynvah (sulopenem etzadroxil and probenecid) for treating certain uncomplicated urinary tract infections...

FDA grants Kind Pharmaceutical an orphan drug designation for AND017 and its use in treating sickle cell disease.

Smiths Medical recalls specific lots of its BLUSelect, BLUgriggs, and BLUperc products due to a manufacturing defect.