
FDA posts a final guidance entitled Early Lyme Disease as Manifested by Erythema Migrans: Developing Drugs for Treatment.

FDA approves an updated label for Eli Lillys Amyvid (florbetapir F 18 injection) for brain imaging to estimate amyloid plaque density in patients bein...
FDA requires and approves updates to the prescribing information for Pfizers Comirnaty Covid-19 mRNA vaccine and Modernas Spikevax Covid-19 mRNA vacci...

A Global Biodefense online post says FDAs sudden decision to halt clinical trials that send samples overseas for processing can hurt the nations biote...
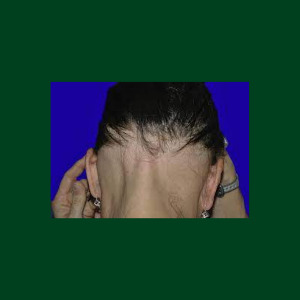
Nektar Therapeutics reports promising results from a mid-stage clinical trial of its experimental therapy rezpegaldesleukin for patients with moderate...
FDA tells healthcare providers to be aware of import alerts for specific Olympus medical devices that are being refused entry into the U.S. due to ong...

FDA says Usona Institute chief medical officer Michael C. Davis is returning to CDER as the Centers deputy director after being gone two and a half ye...

UPenn medical school professor Holly Lynch says there are many questions about the new FDA Commissioners National Priority Voucher program, including ...